Now compare the reactivity of calcium and magnesium with water. Watch the videos above and compare the reactivity of sodium and potassium with water. 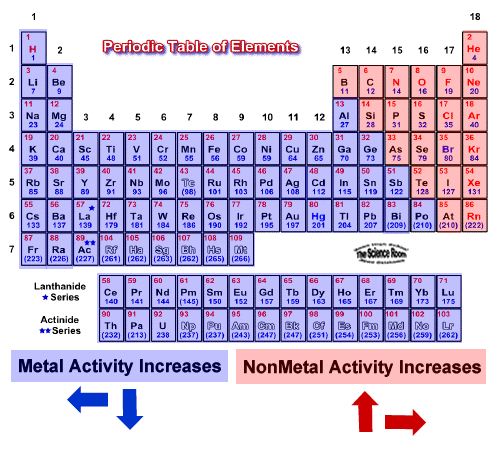
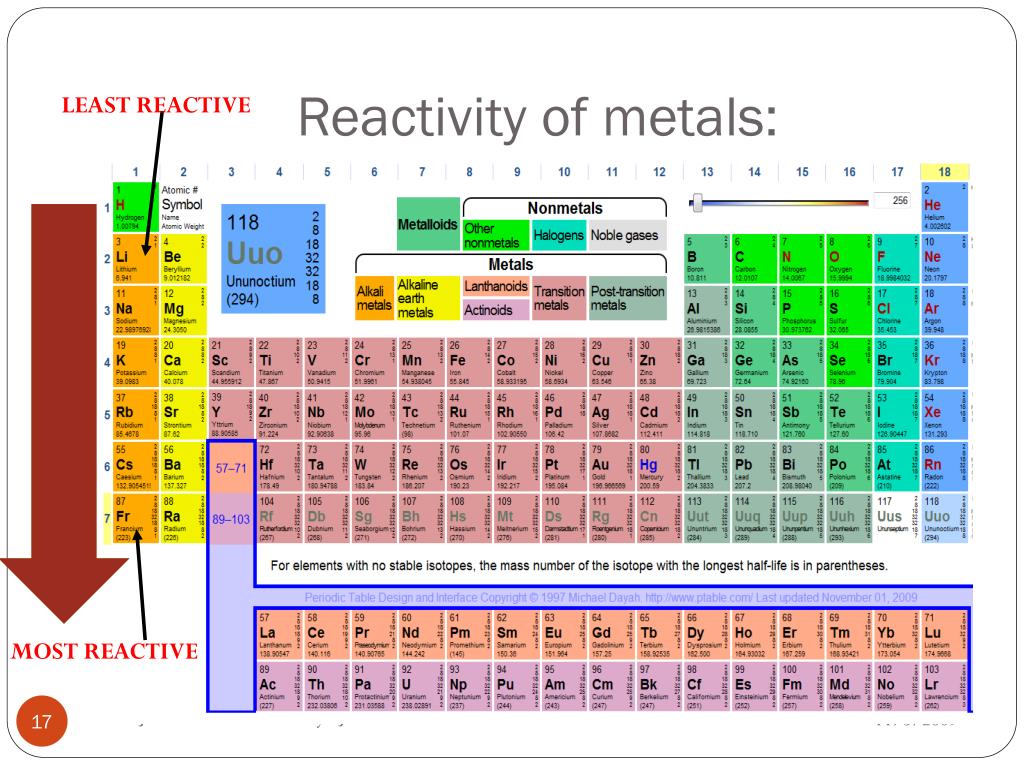
Notice how reactivity of metals increases as we go down a group. Potassium reacts faster than sodium to produce hydrogen gas.Įlements in group 2 also react with water to form hydrogen gas but are not as reactive as group 1 elements. Sodium reacts slowly with water to produce hydrogen gas. Click on the blue writing below to see the reactions. But, reactivity means different things to different chemists, plus it depends on a few factors. So, the most reactive element on the periodic table is either one of these elements. The most reactive metal is cesium, while the most reactive nonmetal is fluorine. These metals react with water to produce hydrogen gas. Cesium is the most reactive metal, while fluorine is the most reactive nonmetal. These metals react readily with other compounds and elements to give away some of their outer shell (valence) electrons. AllĮlements in group 1 are very reactive metals, with the exception of hydrogen Elements in the same group display similar chemical properties and trends emerge as we go along a period and down a group.Īll elements in a particular group share similar chemical properties. The table is divided into groups(columns) and periods(rows). 
At the top of the list are the highly reactive metals that lose electrons during a chemical reaction to form ions. (group 2, or alkaline earth metals) of the periodic table: all group 2 elements have the. The reactivity series of metals is a list of metals arranged in their order of reactivity from highest to lowest. Sodium is an alkali metal, being in group 1 of the periodic table, because it has a single electron in its outer shell that it readily donates, creating a positively charged atomthe Na+ cation. By convention, elements are organized in the periodic table, a structure that captures important patterns in their behavior.Devised by Russian chemist Dmitri Mendeleev (18341907) in 1869, the table places elements into columnsgroupsand rowsperiodsthat share certain properties. The modern periodic table has elements arranged in order of electronicĬonfiguration. Sodium is a soft, silvery-white, highly reactive metal. Recognise that elements can be grouped together according to atomic mass. 
Mendelev was a Russian scientist who was first to Join Seneca to get 250+ free exam board specfic A Level, GCSE, KS3 & KS2 online courses.The periodic table is a way of organizing known elements into groups with Want to learn more about Reactivity Series? Because of this, the metal has to be extracted from the ore (rock) where the metal compound is found. Most metals are only found as compounds because the metal has reacted with other elements in the past. Most metals react with dilute acids to produce a salt and hydrogen gas. Potassium, sodium and lithium all react quickly with cold water to produce a metal hydroxide and hydrogen gas. Platinum, however, is less reactive than copper and so cannot displace copper from a copper sulfate solution. Chromium has values of positive 2,3,4, and 6. Scandium has oxidation state of positive 3. Magnesium + copper sulfate → magnesium sulfate + copper Figure :Transition metals of the first transition series can form compounds with varying oxidation states. The noble gases are almost completely inert. The reactive nonmetals near the metalloids show some incipient metallic character, such as the metallic appearance of graphite, black phosphorus, selenium and iodine. Magnesium is more reactive than copper, so magnesium can displace copper from a copper sulfate solution to create magnesium sulfate. From left to right in the periodic table, the nonmetals can be divided into the reactive nonmetals and the noble gases. the MOST reactive family of METALS - all members are silver-gray in color, soft, have luster and ONE valence electron. A displacement reaction happens when a more reactive metal (one that forms positive ions more easily) displaces a less reactive metal from a compound. Group 18 - the LEAST reactive elements on the periodic table - all have 8 valence electrons, except for Helium (which has one energy level and is stable with 2 valence electrons). This is called a displacement reaction.Ī metal can only displace another metal from a compound if it is located above it in the reactivity series. These are called native metals.Ī more reactive metal (one that forms positive ions more easily) can displace a less reactive metal from a compound. Very unreactive metals, such as gold and platinum, are found in the Earth’s crust as pure metals. The group 17 elements are highly reactive, colorful nonmetals. For example, the group 1 elements are all soft, reactive metals. Metals can be arranged in order of their reactivity. A periodic table group is a column, while a periodic table period is a row. The easier it is for a metal to form its positive ion, the more reactive the metal is. When metals react with other substances, the metal atoms always form positive ions.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
